usp class vi elastomers
Isolast Plus J9515 J9516 - Superior sealing with FDA USP class VI and SP 3-A compliance. Who uses USP Class VI elastomers.
Ia USP Class VI andor ISO 109933 will be required.

. All these special grade products have passed this rigorous test. For more information on UPS Class VI seals or to place your order contact Eastern Seals by phone 44 0 1670 840529 or email saleseasternsealscouk. Colorites Cellene line of thermoplastic elastomer compounds is an alternative to PVC phthalate-based plasticized compounds and various rubber materials in medical devices medical packaging and other regulated markets.
Master Bond systems are very versatile and can be used for both disposable and. These medical silicones come in thicknesses between 005 and 250 and in the following durometers. Sil 714002 USP class VI Silicone 1 70 Yes transl.
The FDA USP Class VI silicones that JBC sources are translucent in color and supplied as compression-molded sheet stock or continuous rolls. Pharmacopeial Convention USP is a non-profit organization with a purpose of creating standards for medications food ingredients dietary supplements and healthcare technologies. The USP publishes bio compatibility protocols for the plastics and polymers used in medical devices or surgical equipment that may come in.
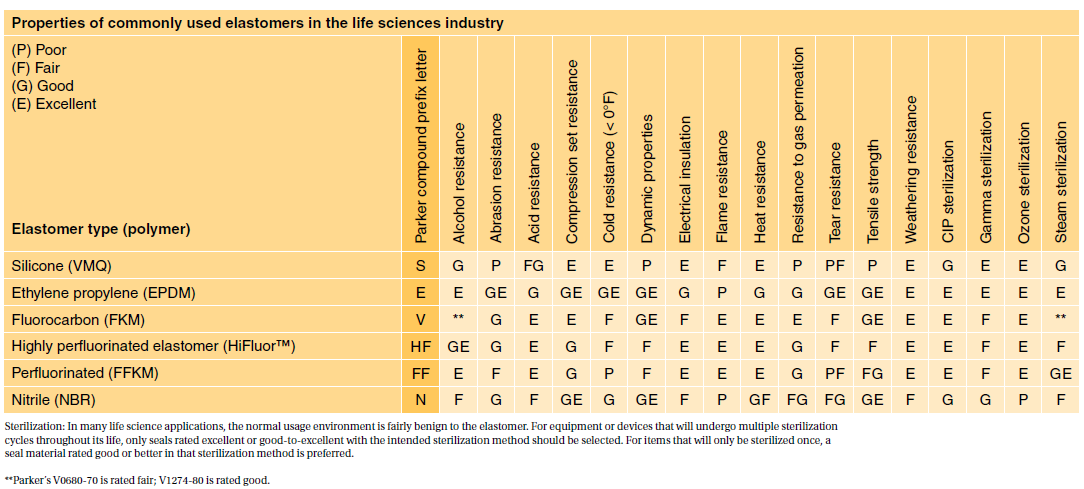
Testing is commonly done as per USP which requires three types of. Most applications are fairly benign to elastomers. Please contact the division for assistance in selecting materials in these situations.
Thermoplastic Elastomers for Medical Devices Meet USP Class VI and ISO 10993 Standards. Class VI elastomers are highly sought after by those in the medical and pharmaceutical industries. USP Class VI Approved Medical Grade Plastic Materials - USP - US.
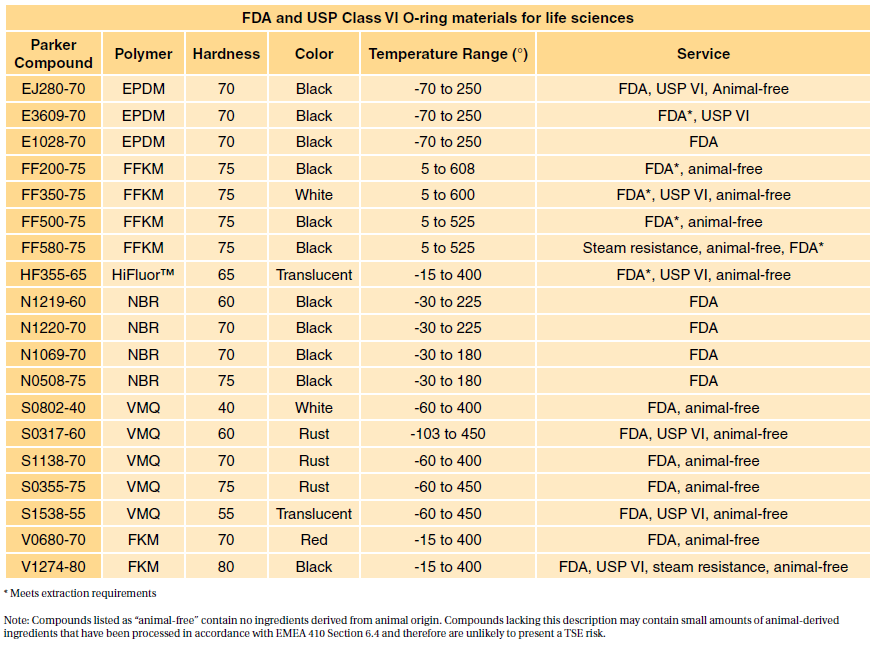
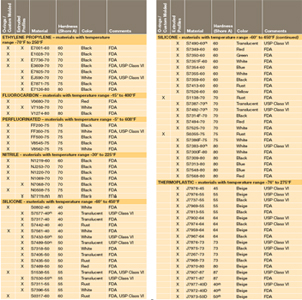
Table 1 shows our standard programme FDA compliant com- FDA and USP class VI compliant. 10 20 30 40 50 60 70 and 80. Liveo Class VI Elastomers C6- eries are a series of one-part uncatalyzed silicone elastomer raw materials.
Specialty Silicone Products SSP makes USP Class VI silicones for medical applications. UV10Med offers very low viscosity in comparison to UV10TKMed which offers moderate yet flowable viscosity. Moulded O-rings class 1 less than 10 furnace black These can be produced in all possible dimensions up to diameter 1400 mm internal.
Sil 714001 USP class VI Silicone 1 70 Yes transl. The Premium Package includes all elastomers in the wetted path of the SLA Series Biotech including O-rings and Valve Seats are USP Class VI and ADI Free and include a full package of certificates. Find Usp Approved Elastomers related suppliers manufacturers products and specifications on GlobalSpec - a trusted source of Usp Approved Elastomers information.
Additionally 22 Material Certifications certifying the composition of all materials in the wetted path and International Calibration. FDA and USP Class VI O-Rings. In addition UV10TKMed also meets the ISO 10993-5 specifications.
Two component EP21AOLV-2Med passes USP Class VI biocompatibility testing and ISO 10993-5 cytotoxicity requirements. Specially formulated for long term sealing. USP Class VI and FDA u001c White Listu001d Silicone and Organic Elastomer Compounds for Healthcare Products.
Valley Seal offers a wide range of USP Class VI and FDA elastomeric materials for the Healthcare Industry. It resists many chemicals including water oils fuels and. Sanitary diaphragm valves have USP Class VI diaphragms.
Any company wishing to produce USP Class O-rings for any kind of food. 7 USP Class VI materials. These materials can be supplied as precision extruded and cut seals used as static face or static radial seals long length.
Tire Rubber Elastomer Products 3011-TR Touchscreens Graphic Overlays Membrane Switches 3575-TS Trade Show Display Events 2542-TD Trading Companies. However some applica-tions such as implantable devices are extremely complicated. As a result any rubber compounds certified under USP Class VI are proven to have high levels of biocompatibility meaning they are ideally placed to use in medical and pharmaceutical situations.
Sanitary pumps require Class VI O-rings and seal material. USP Class VI testing is conducted by producing an extract of the product with different extraction fluids such as polyethylene glycol and vegetable oil and injecting it in specimen rabbits and mice in vivo alive to observe the biological response to the extract. These materials may be blended if desired to achieve intermediate hardnesses.
JBC can also source materials in custom colors but the pigments. What are Class VI elastomers. USP Class VI and FDA White List Silicone and Organic Elastomer Compounds for Healthcare Products.
June 15 2017 By Lee Goldberg. Pharmacopoeia Class VI judges the suitability of plastic material intended for medical device manufacturers. The SSP2390 family of products isnt new but the marketplace for these materials continues to evolve.
Both products are optically clear and they meet the USP class VI specifications. A workshop Modernization of USP Packaging Standards for Glass and Elastomeric Components will take place June 1920 2017 at the USP Meetings Center in Rockville Maryland to discuss the proposals for three new chapters including this one Elastomeric Closure Functionality in Injectable Pharmaceutical PackagingDelivery Systems 382 and. USP Class VI elastomers.
Has a full range of specialty adhesives epoxies primers for polyolefins UV curables and silicones that have been fully tested to meet USP Class VI requirements. Darcoid and Parker offer a wide range of USP Class VI and FDA. The cross-linked molecular chains enable them to combine the resilience and sealing force of an elastomer with the chemical inertness and thermal stability of PTFE.
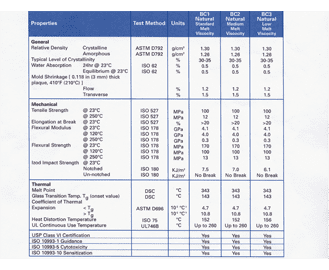
The resulting elastomers range in hardness from soft to firm nominally 35 to 65 shore A durometer. There are two important organisations that play a key role in the regulation of elastomers and O-rings these being the United States Pharmacopeia USP and the US Food and Drug Administration FDA as well as meeting European regulation EU1935. To keep up with the changes medical device designers and manufacturers need elastomers they can trust.
Category Featured Company Information. USP Class VI materials EPDM Silicone Fluorocarbon and Perfluoroelastomer 24 materials which are compliant to FDA 21 CF R1772600.

Iso 10993 Vs Usp Class Vi Medical Molding And Bicompatible Rubber The Rubber Group

Usp Class Vi What Is It And How Does It Apply To Elastomers Barnwell

O Rings Fda And Usp Class Vi Darcoid Rubber Company Oakland California

Biopharmaceutical Usp Class Vi Gaskets Newman Sanitary Gasket
Usp Class Vi Standard Anderson Negele North America

Why You Need Certified Usp Class Vi Silicones Specialty Silicone Products Inc
Fda Usda Nsf51 Usp Class Vi Compliant Seals Products

Usp Class Vi Standard Anderson Negele North America

Usp Class Vi Seals Compliant To Food Grade Standards Barnwell

What Is Usp Class Vi Testing Tbl Plastics

Iso 10993 Vs Usp Class Vi Medical Molding And Biocompatible Rubber
![]()
Medical Silicones Specialty Silicone Products Inc

Parker Medical Fda O Rings Sealing Devices
![]()
Medical Silicones And Usp Vi Biological Reactivity Tests In Vivo Jbc Technologies
![]()
Specialty Silicone Products Usp Class Vi Silicones Today S Medical Developments

Usp Class Vi What Is It And How Does It Apply To Elastomers Barnwell